Halogens role in volcanic sulphur oxidation: photochemical modelling and isotopic constraints (In preparation)
Abstract
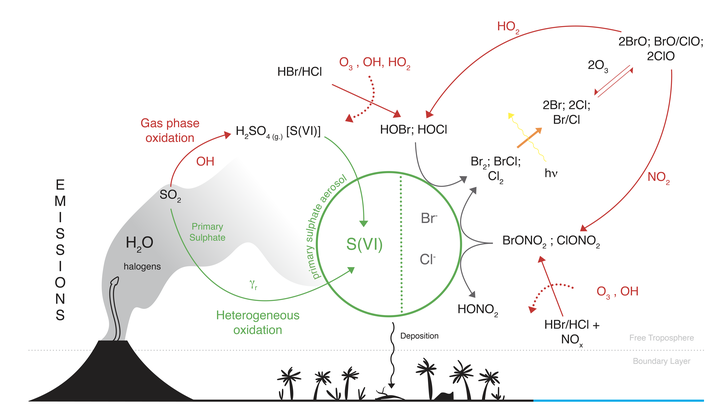
The photochemical box-model CiTTyCAT is used to simulate volcanic sulphur oxidation and the resulting volcanic sulphate oxygen isotopic composition within the core of volcanic plumes. The focus is on volcanic halogens role in volcanic sulphur chemistry during small volcanic eruptions or passive degassing. The model accounts for SO$_2$ oxidation by OH in the gas phase, and by H$_2$O$_2$, O$_3$, O$_2$ catalysed by transition metal ions (TMI), and HOX (HOX = HOBr + HOCl) in the liquid phase of either volcanic water droplets (referred here as condensing plume), or sulphate aerosols (referred here as non-condensing plume). It also describes transfer of oxygen mass-independent anomalies (O-MIF) from atmospheric oxidants to sulphate. For condensing plumes (liquid water), the first finding is that ozone depletion events (ODEs) triggered by halogens heterogeneous chemistry may also occur in plumes cores in presence of water droplets. The second finding is that, halogens presence does not affect the hierarchy of oxidation pathways. The O$_2$/TMI sulphur oxidation pathway remains dominant even for relatively low TMI aqueous concentrations, generating volcanic sulphates with low O-MIF (~ 0 ‰), this result is comparable to isotopic measurements on sulphates collected from volcanic ash-deposits. For non-condensing plumes (only sulphate aerosols), the first finding is that ODEs can occur even at very low halogen loadings (~50 ppbv). The rate of sulphur oxidation is much lower than for condensing plumes, and sulphur oxidation is dominated by H$_2$O$_2$ on sulphate aerosols and by OH in the gas phase. The second finding is that, the relative contributions of H$_2$O$_2$ and OH is determined by halogens loading, and that the higher the halogens concentration is, the higher is the H$_2$O$_2$ contribution to sulphate production. The third finding for non-condensing plumes is that halogens presence does not impact significantly the final sulphate O-MIF, which remains between the range of 0.8 - 1.4 ‰. The latter result is in contrast with the lack of O-MIF measured in sulphates collected from volcanic ash, suggesting different hierarchy of oxidation pathways might be active for different plume environments. The condensing and non-condensing simulations results suggest that the sulphate found on volcanic ash-deposits is likely to be formed via SO$_2$ oxidation in water-rich phases and not in the gas phase or on sulphate aerosols during passive degassing.